About NIH
The National Institutes of Health (NIH), (under the U.S. Department of Health and Human Services) is the primary U.S. agency dealing with, and the largest source of funding for, medical research in the world. NIH is made up of 27 Institutes and Centers, each with specific research agendas and interests often focusing on particular diseases or body systems.
Researchers from Canadian universities are eligible for many NIH funding opportunities. Please consult OSR staff early for more information on submitting to the NIH.
The information below outlines McGill University’s requirements for all Investigators applying for, holding, or working on studies that must comply with the US Public Health Service Financial Conflict of Interest Regulations (PHS FCOI Regulations). The PHS FCOI Regulations establish Investigator reporting and training requirements and apply to all PHS-funded research, including NIH-funded research.
Submission information: Annual grant renewal, update on awarded grant
Before submission of a grant application, on annual grant renewal, and within an update on awarded grant, the McGill PI must ensure the following:
- Provide a List of Investigators. This list must include all McGill Investigators, as well as any Subrecipient Investigators involved with the study;
- Ensure that all McGill Investigators have signed and submitted a McGill Significant Financial Interest Disclosure form;
- Ensure that an authorized official of the Subrecipient has signed and submitted to McGill the Subrecipient Commitment Form for NIH. Please contact OSR to obtain the Subrecipient Commitment Form. Where a Subrecipient/External Investigator opts to follow McGill FCOI Policy, they must also submit a McGill Significant Financial Interest Disclosure form.
Before submission of a grant application and every 4 years
The McGill PI and all Investigators following McGill FCOI Policy must complete the NIH FCOI tutorial. The NIH Financial Conflict of Interest tutorial was designed by the National Institutes of Health to provide education training on what constitutes a financial conflict of interest. It provides guidance on the proper policies and practices regarding these issues and is required for anyone involved with an NIH-funded project. Please visit the NIH’s website to take the online tutorial.
Information regarding Financial Conflict of Interest
Where Subrecipient/External Investigators who are following their institutions’ FCOI Policy are found to have a FCOI, the Subrecipient institution must report to McGill the FCOI before awarded funds are released, annually, and within 30 days of a new FCOI being identified. Please see McGill’s procedure on the US Public Health Service Financial Conflict of Interest Regulations here.
Two-Factor Authentication (2FA) required for all eRA Commons, Internet Assisted Review (IAR) and ASSIST users
As of September 15, 2021, the National Institutes of Health (NIH) will require two-factor authentication (2FA) to access eRA Commons, Internet Assisted Review (IAR) and ASSIST.
2FA is a form of multi-factor authentication that helps verify you are the person who is logging into your account. Activating 2FA on your account reduces the risk of cyber-attacks such as phishing, malware, and other attempts to take over your account.
Follow the steps outlined in this email, sent out to users by the eRA Commons system on June 15th, to enable 2FA. The graphic below summarizes the key steps of the process:
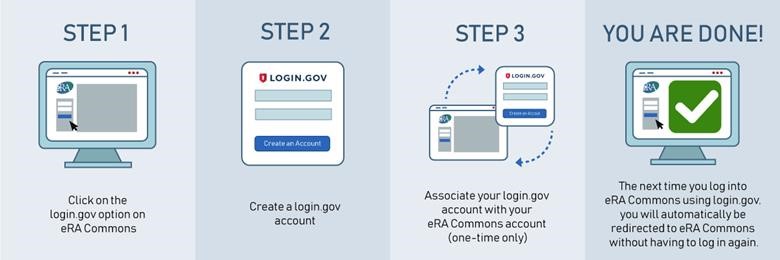
Setting up 2FA for eRA Commons will be easier if you have already opted into McGill 2FA. If you have not yet enabled 2FA on your McGill account, go to www.mcgill.ca/2fa to enroll and complete the setup, which takes approximately 10-15 minutes. It is recommended to do so during business hours (Monday-Thursday, 8 am to 7 pm, Friday 8 am to 5 pm) so that you can get IT support if you need it.
For questions or assistance with McGill’s 2FA, contact the IT Service Desk at www.mcgill.ca/itsupport/servicedesk.
