
Professor
B. Sc. (University of Zagreb, Zagreb, Croatia, 2001)
Ph. D. (University of Iowa, Iowa City, Iowa, USA, 2006)
Postdoctoral fellow (Pfizer Institute for Pharmaceutical Materials Science, Cambridge, UK, 2006-2008)
Herchel Smith Research Fellow (University of Cambridge, Cambridge, UK, 2008-2010)
Awards
2019 Award for Research Excellence in Materials Chemistry, Canadian Society for Chemistry
2018 National Research Council Steacie Prize for Natural Sciences
2018 Member of the College of New Scholars, Artists and Scientists of the Royal Society of Canada
2018 Royal Society of Canada Rutherford Memorial Medal in Chemistry
2018-2019 World Economic Forum Young Scientists member
2017 NSERC E. W. R. Steacie Memorial Fellowship
2017 NSERC Discovery Accelerator
2016 McGill University Principal’s Award for Outstanding Emerging Researchers
2015-2019 McGill University Dawson Research Scholar
2015 McGill University Tomlinson Scientist Award
2014 RSC ChemComm Emerging Investigator Award and Lectureship for Organic Chemistry
2013 Fellow of the Royal Society of Chemistry (RSC, UK)
2011 RSC Harrison-Meldola Memorial Award for "developing solid-state methodologies which explore and combine new types of molecular self-assembly"
Contact Information
Office: P&P 104
Phone: (514)398-3959
Email: tomislav.friscic [at] mcgill.ca
Lab: P&P 028
Lab Phone:
Web Page: Friscic Group Website
Research Themes
- Materials Chemistry
- Synthesis/Catalysis
Research Description
Mechanochemistry and solid-state chemistry for green chemical synthesis
Coventional chemical processes are based on solution chemistry. As a result, laboratory- and industrial-scale chemical reactions are inherently wasteful of solvent (which is an obvious neccessity for solution-based reactions) and of energy (which becomes dissipated in the large volume of solvent).
Our research is inspired by the idea that solid-state reactions will provide cleaner, faster and more diverse chemical reactions. This is elaborated in a recent highlight: "New opportunities for materials synthesis using mechanochemistry" J. Mat. Chem. 2010, 20, 7599-7605.
Whereas some solid materials will readily undergo solid-state reactions, most of them do not. For this purpose we are developing mechanochemical approaches, in which grinding together of reactants with catalytic additives results in the self-assembly of advanced materials, such as porous metal-organic frameworks (MOFs), directly from the simplest and cheapest possible precursors. These advanced mechanochemical methodologies include:
- liquid-assisted grinding (LAG) and
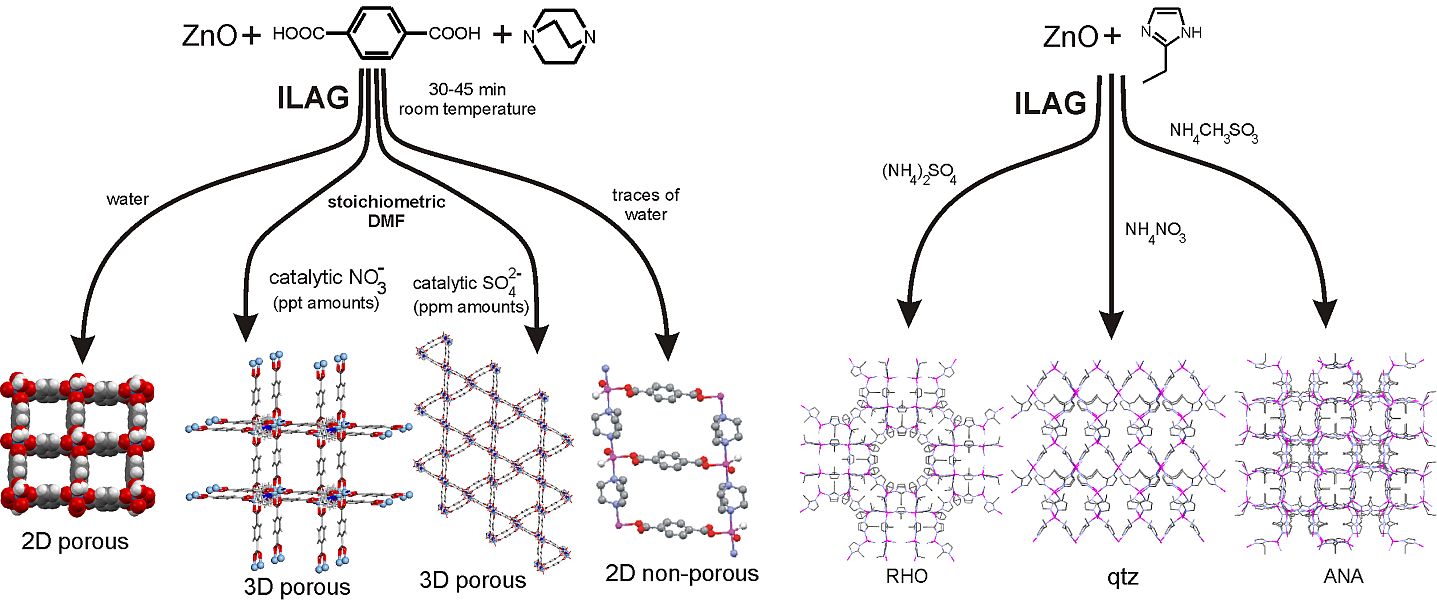
- ion- and liquid-assisted grinding (ILAG)
These methodologies allow conducting chemical reactions independent on the solubility of the reactant materials and lead to overall 1000- or 10000-fold reductions in the use of solvent and energy compared to conventional processes.


Besides enabling cleaner and faster reactions, the simple catalytic salts used in ILAG also provide an exciting opportunity to direct the topology (pore shape and size) of porous MOFs and thus discover potential future catalysts, ion exchangers, conductors and gas storage materials for fuel gases, such as methane or hydrogen, or for waste gases, such as carbon dioxide.
In addition to discovering new approaches to mechanochemistry, and studying the underlying mechanisms of solid-state reactivity ("mechanisms of mechanochemistry"), the research directions which are pursued in our group include:
- Energy- and solvent-free reactions in which presumably "inert" materials are activated so as to undergo chemical reactions under mildest possible conditions.
- Organic reactions using mechanochemistry: we are investigating reversible organic reactions and the coupling of mechanochemistry with other forms of solid-state reactivity as a means to conduct multi-step organic synthesis reactions exclusively in the solid state.
- Metallopharmaceuticals and metallodrugs: metal-organic materials with pharmaceutical relevance. By using mechanochemistry, we have recently demonstrated an inexpensive route to transform bismuth oxide into bismuth subsalicylate - the active ingredient of the drug Pepto-Bismol.
For more information, check out our latest papers on
Ion- and liquid-assisted grinding:
- "Rapid room-temperature synthesis of zeolitic imidazolate frameworks using mechanochemistry" Angew. Chem. Int. Ed. 2010, 49, 9640-9643
- "Towards an environmentally-friendly laboratory: dimensionality and reactivity in the mechanosynthesis of metal-organic compounds" Chem. Commun. 2010, 46, 9191-9193.
- "Ion- and liquid-assisted grinding: improved mechanochemical synthesis of metal-organic frameworks reveals salt inclusion and anion templating" Angew. Chem. Int. Ed. 2010, 49, 712-715. Highlighted as an Angewandte Chemie "Hot Paper"
Organic mechanochemistry:
Reversible covalent synthesis using mechanochemistry: solid-state product selection is different than in solution" Chem. Sci. 2011, 2, 696-700.
Metallopharmaceuticals:
"Mechanochemistry of magnesium oxide revisited: a facile route to derivatise pharmaceutical ingredients by liquid-assisted grinding (LAG)" Chem. Commun. 2010, 46, 6368-6370
Currently Teaching
