Seawater: memory keeper, energy source, and pollution tracking

Sampling seawater just below the surface of a seagrass bed in Quatsino Sound, British Columbia. Credit: Mike McDermid
What can a bottle of seawater tell you about the fish living below?
Seawater holds “memories” in the form of DNA from fish and invertebrates that have recently passed by. This information, called environmental DNA or eDNA, can be used by scientists to track species across space. This new approach is being used by researchers like McGill Professor Jennifer Sunday and her colleagues at the Pacific eDNA Coastal Observatory (PECO) to track “biogeography” like we forecast the weather. The PECO network has been collecting seawater in bottles from Juneau, Alaska to San Diego, California to find out which fish live where and how these change over time, focusing on seagrass habitats across this large coastal region. With this information, the researchers will survey the geographic distributions of hundreds of fish and gain a better understanding of how species live together in different environments – as potential consumers, competitors, and invasive species – all from bottles of water. “Networks like PECO could mark the start of a new frontier in tracking underwater biodiversity. Groups like these are forming around the world,” says Sunday, who is Assistant Professor in the Department of Biology.

Detecting water pollutants in a blink of an eye
Imagine using your mobile phone to track the spread of water contaminants – such as oil spills or even viruses like COVID-19 – in the blink of an eye. Researchers from McGill University have developed new artificial intelligence to make the invisible visible using advances in lasers, optics, and mobile technology. The new technology uses remote sensing, meaning it acquires information from a distance, and could one day be deployed on satellites to detect in real-time pollutants as small as a nanometer to a centimeter in the water systems around the globe. “This information could be used to identify, predict, and address the spread of environmental contaminants and outbreaks of diseases or viruses,” says Professor Parisa Ariya of the Department of Chemistry and Department of Atmospheric and Oceanic Sciences, who led the team of researchers. “It’s a game-changer, allowing governments, industries, and communities to act quickly, share information, and reduce damage to the ecosystem and risks to public health.”
"Novel Dynamic Technique, Nano-DIHM, for Rapid Detection of Oil, Heavy Metals, and Biological Spills in Aquatic Systems" by Ryan Hall et al. was published in Analytical Chemistry.
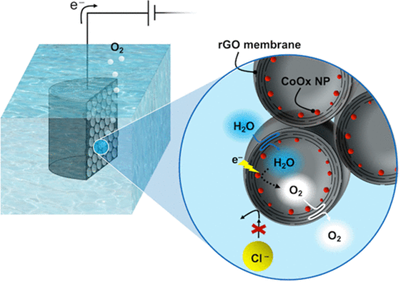
The researchers used X-ray contrast imaging at the Canadian Light Source at the University of Saskatchewan to confirm that the structure of the graphene oxide scaffold with closed graphene oxide pores were able to enclose the catalyst and prevent it from being destroyed.
Extracting hydrogen from seawater
In principle, seawater offers an ideal source of hydrogen for clean energy. But, in practice, the electricity used to extract the hydrogen from seawater produces unwanted by-products (from the salt in the water) that poison the catalyst and shut down the process. By using stacked graphene sheets (sheets of carbon atoms similar to those in pencil lead, each as thin as a thousandth of a hair), a team of researchers, led by McGill professor Marta Cerruti of the Department of Materials Engineering, have now built a porous, three-dimensional, electrode in the form of a sponge that has successfully separated the water from the unwanted chemical compounds. The next challenge will be scaling up the process to mass produce the electrodes, which could also potentially be used for other reactions where it is important to prevent interference from certain molecules.
"Selective Catalytic Electro-Oxidation of Water with Cobalt Oxide in Ion Impermeable Reduced Graphene Oxide Porous Electrodes" by Gabriele Capilli et al. was published in ACS nano.
About McGill University
Founded in 1821, McGill University is home to exceptional students, faculty, and staff from across Canada and around the world. It is consistently ranked as one of the top universities, both nationally and internationally. It is a world-renowned institution of higher learning with research activities spanning three campuses, 11 faculties, 13 professional schools, 300 programs of study and over 39,000 students, including more than 10,400 graduate students.
McGill’s commitment to sustainability reaches back several decades and spans scales from local to global. The sustainability declarations that we have signed affirm our role in helping to shape a future where people and the planet can flourish.

