Novel gene implicated in osteoporosis

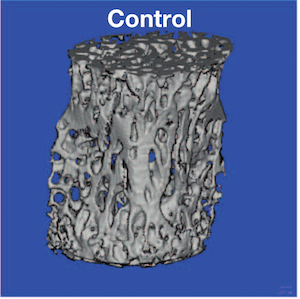
PHOTO: CT image shows lack of bone in a mouse with the key gene (EN1) removed”
Using extensive genetic data compiled by the UK10K project, an international team of researchers led by Dr. Brent Richards of the Lady Davis Institute at the Jewish General Hospital has identified a genetic variant near the gene EN1 as having the strongest effect on bone mineral density (BMD) and fracture identified to date. The findings are published in the forthcoming issue of the prestigious journal Nature.
“EN1 has never before been linked to osteoporosis in humans, so this opens up a brand new pathway to pursue in developing drugs to block the disease,” Dr. Richards, an Associate Professor of Medicine at McGill University, says by way of explaining the importance of the discovery.
“The effect of this uncommon genetic variant that we identified in this gene is twice as large as any previously identified genetic variants for BMD and fracture,” adds Vince Forgetta, first author on this collaborative project from the Genetic Factors for Osteoporosis Consortium, and a Research Associate at the Lady Davis Institute.
Osteoporosis is a common disease that will lead to fractures in between one-third and one-half of all women over the course of their lives. Because osteoporosis becomes more severe with age, it is becoming more prevalent with the overall aging of the population. There are currently few safe and effective treatments for osteoporosis, and no curative therapies available.
The UK10K project has measured genetic variations in 10,000 individuals in great detail, allowing researchers to correlate rare genetic changes with human disease by comparing the DNA of healthy individuals with those who have health problems. The use of such an extensive sample size allows for the observation of genetic variants that are not discernable among smaller groups. This particular study also stands as proof of principle that uncommon genetic variants can have a significant impact on common diseases.
“The hypothesis is that the genetic sequencing being done by UK10K will expose previously unknown genetic factors underlying disease,” said Dr. Celia Greenwood, a biostatistician who is Senior Investigator at the Lady Davis Institute and Associate Professor at McGill University, and co-chaired the statistics group for a companion Nature article on the methodology behind UK10K. “We are finally able to extract enough data to discern variants that are rare in the overall population and are more frequent among those with common diseases. This is precisely what has been revealed in the case of EN1 and osteoporosis.”
This study represents an initial realization of the hope that accompanied the development of genetic sequencing technology: that sophisticated analysis of the genome would reveal those genes associated with disease. The promise for the contribution genetics can make to human health lies in the discovery of novel compounds that can counter the effect of deleterious genetic variants influencing these genes.
“Whole-genome sequencing identifies EN1 as a determinant of bone density and fracture” by J. Brent Richards et al, Nature, on-line September 14, 2015.
See also, “The UK10K project identifies rare variants in health and disease,” Nature, September 14, 2015.
For further information, and to arrange interviews, contact:
Tod Hoffman
Research Communications Officer
Lady Davis Institute
Tel.: 514-340-8222 x 8661
Cell: 514-433-3500
Email: thoffman [at] jgh.mcgill.ca
For more about the Lady Davis Institute: www.ladydavis.ca
For more about the Jewish General Hospital: www.jgh.ca