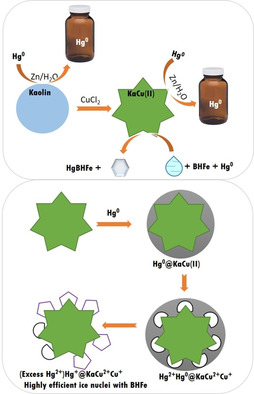
Kaolin, a natural and inexpensive clay mineral, is ubiquitous in soil, dirt, and airborne particles. Amongst four commonly available clay minerals, kaolin, as a result of its layered structure, is the most efficient natural gaseous Hg adsorbent to date (Langmuir maximum adsorption capacity Qm=574.08 μg g−1 and Freundlich Qm=756.49 μg g−1). The Hg uptake proceeds by homogeneous monolayer and heterogeneous processes. Hg physisorption on kaolin occurs in the dark, yet the adsorption rate is enhanced upon irradiation. The effects of several metal complexes, salts, halides and solvents on the Hg uptake were examined. The addition of CuCl2 particles leads to a significant enhancement of the Hg uptake capacity (>30 times) within second timescales and without irradiation. The physisorption with kaolin is switched to chemisorption upon the addition of CuCl2 to kaolin. This process is entirely reversible upon the addition of Zn/Sn granules at room temperature without any added energy. However, the investment of a small amount of renewable energy can speed up the process. This technology demonstrates the facile and efficient capture and recycling of elemental Hg0 from air. A wide range of metal particles and diverse physicochemical processes, which include the microphysics of nucleation, are herein examined to explore the potential reaction mechanism by using a suite of complementary analytical techniques. These new mechanistic insights open a new era of energy‐neutral environmental remediation based on natural soil/airborne particles.
To read more, please consult CHEMSUSCHEM journal (volume 12, issue 23, 2019) or visit https://onlinelibrary.wiley.com/doi/abs/10.1002/cssc.201902955
Authors: Dr. Mainak Ganguly , Yuanyuan Tao, Bryan Lee, Prof. Parisa A. Ariya
Area of Research: Atmospheric Chemistry
Dr. Ganguly before moving to a new position with a prestigious institution in India, was a postdoctoral researcher in our department. His research was related to design of sustainable nanomaterials (surrogates for mineral dust), ice nucleation and its environmental aspects, preparing of nano trap for environmental cleanup, green chemistry etc.
Prof. Parisa Ariya is a James McGill Professor of Atmospheric and Interfacial Chemistry, Professor at the Department of Atmospheric & oceanic Sciences as well as the Department of Chemistry at McGill. Her laboratory's research explore major fundamental, and applied research questions on chemical and physical processes involving aerosols, as well as organic and metal pollutants of relevance to the Earth's atmosphere and its interfaces. Our direct research contributions are to the fields of atmospheric sciences and climate change, specifically aerosol-cloud interaction, air pollution, atmosphere-ocean interactions, physical and analytical chemistry, sustainable chemistry and technology, nanoscience, environmental health and medicine.