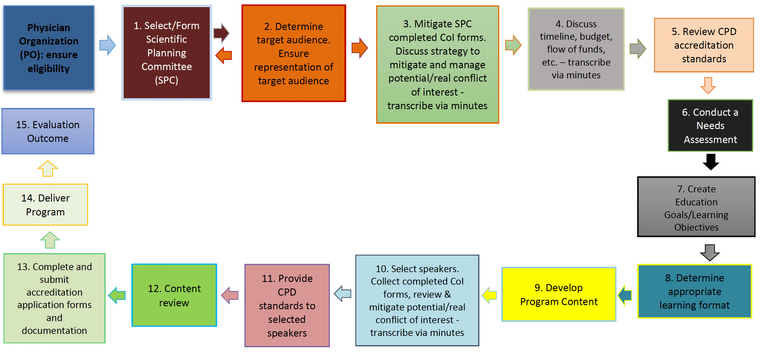
In compliance with the National Standard for Support of Accredited CPD Activities, and to safeguard attaining CPD credits for your activity, please ensure to follow the planning process described below:
Physician Organization (PO): Ensure eligibility
(Click on the image to enlarge it)
- Select/Form Scientific Planning Committee
- Determine target audience. Ensure representation of target audience
- Mitigate SPC completed CoI forms. Discuss strategy to mitigate and manage potential/real conflict of interest - transcribe via minutes
- Discuss timeline, budget, flow of funds, etc. – transcribe via minutes
- Review CPD accreditation standards
- Conduct a Needs Assessment
- Create Education Goals/Learning Objectives
- Determine appropriate learning format
- Develop Program Content
- Select speakers. Collect completed CoI forms, review & mitigate potential/real conflict of interest - transcribe via minutes
- Provide CPD standards to selected speakers
- Content review
- Complete and submit accreditation application forms and documentation
- Deliver Program
- Evaluation Outcome
Activities must be developed or co-developed with a Physician Organization (PO). The PO must be involved in the development of the CPD activity from appointing the SPC members to the evaluation outcome.
Following the ‘Infographic - CPD Activity Planning Process Overview’ will ensure the activity is developed, planned and implemented according to the ethical, educational, and administrative standards as directed by CPD/CME regulatory authorities.
- Appointing the Scientific Planning Committee (SPC) must be done in collaboration with the PO. If you are developing an activity that will be held in Quebec, ensure to engage two Committees: Scientific Planning Committee (SPC) & Organizing Planning Committee (OPC): Scientific Planning Committee (SPC): Chair, Selecting Members and Responsibilities; Composition of the SPC and OPC
- It is mandatory that representatives of the target audience be included in the SPC. If the target audience is interprofessional, the SPC must include representation from each profession. If the activity will be held in Quebec, certifying to engage two committees: Scientific Planning Committee (SPC) & Organizing Planning Committee (OPC): Composition of the SPC and OPC. The SPC may also include non-Canadian members if the results of the needs assessment indorse the need.
- The SPC must discuss and record in Meeting Minutes key elements linked to the development, planning and implementation of the activity. Discussions must include: the method used to review the completed Declaration of Potential Conflict of Interest (CoI) Forms, including those of the SPC members; Strategy Used to Manage Potential or Real Conflicts of Interest.
- The SPC must discuss and record in Meeting Minutes the activity timeline, Budget, and the flow of funds. The SPC must ensure that the funds received from for-profit and/or not-for-profit are in the form of an educational grant payable to the SPC/PO. The SPC/OP are accountable and responsible for the disbursements of funds. NOT PERMITTED: Influence from industry (for-profit and/or not-for-profit entities) over any of the planning aspects of an activity is not permitted.
- To facilitate the accreditation/certification process, it is highly recommended that the SPC/PO consult the CPD standards established by CPD regulatory authorities: National Standards - Accredited CPD Activities; CMA Guidelines - Physicians in Interactions with Industry; CFPC - Understanding Mainpro+ Certification; Royal College - Accreditation Toolkit; CQDPCM - Quebec Code of Ethics.
- The SPC must conduct a Needs Assessment to determine the current standing of the target audience’s knowledge and identify gaps necessary to develop, plan and implement the activity.
- The SPC must develop the Learning Objectives that address the identified gaps/needs of the target audience. L.O.s must be developed based on the results of the needs assessment conducted; NOTE: It is essential that L.O.s be written from the learner’s perspective and that they clearly state what a participant will know or be able to do as a result of attending an activity or session;
- The SPC must ensure that the selected learning format/delivery of the content is consistent with the identified learning gaps/needs and learning objectives. Learning formats include: conferences, workshops, online modules, webinars, etc.
- The activity topics and content must be based on the data collected from the needs assessment and the gaps identified. In addition, concise learning objectives for each topic must clearly classify the intent of the educational session. The SPC is also responsible to ensure that content presented avoids copying images, videos, tables, cartoons, and graphs from copyright publications. Copyright Criteria for PowerPoints
- Speakers should be selected based on the expertise on the content being presented and should have minimal conflict of interest. The SPC must ensure that all resource persons, including the SPC members, communicate (verbal and with slides) a statement on Conflict of Interest to the audience.
- The SPC must ensure that all resource persons delivering CPD content (i.e. speakers, moderators, facilitators, etc.) are informed of CPD accreditation/certification standards. Quick Tips for Speakers
- The SPC is responsible to review the content prior to the activity start date.
- The SPC is accountable for the accuracy and transparency of the information provided on the CPD Accreditation/Certification Application Form and responsible for Submitting Mandated Supporting Documentation.
- The SPC must ensure that the educational content delivered is balanced, scientifically valid and evidence based.
- Appropriate evaluation (post-activity) outcome should be reviewed by the SPC. As such, no later than eight (8) weeks following the completion of the activity, the SPC Chair should complete and submit a Final Report Form and supporting documentation to finalize the accreditation/certification process.